How will gene therapy impact the future of bleeding disorders?
08/31/2023
About 30,000 Americans are living with the rare bleeding disorder, Hemophilia B. Thanks to the recent FDA approval of Hemgenix®, the first-ever gene therapy to treat a bleeding disorder, some of these patients have the potential to alleviate significant burdens associated with traditional factor replacement treatments.
Dr. Amy Shapiro—pediatric hematologist and CEO and co-founder of the Indiana Hemophilia & Thrombosis Center (IHTC) in Indianapolis—shared insight into what this news can mean for eligible patients. Dr. Shapiro and her IHTC colleagues have had experience in gene therapy, participating in clinical trials and research. IHTC will soon be able to administer the treatment to patients who qualify—if they decide the treatment is right for them.
Q: What does this new gene therapy treatment mean to eligible patients with Hemophilia B?
Dr. Shapiro: We’ve seen gene therapy’s success in other areas of medicine, and this is the first FDA-approved gene therapy for bleeding disorders. The success of this new treatment means the future is bright for the development of more gene therapies for individuals with bleeding disorders.
Hemophilia impacts patients differently, and many of these patients—especially those in the severe range—can experience life-altering bleeds. These patients often go through frequent, time-consuming factor replacement therapies at home. They might make frequent visits to the emergency department and doctor’s office, all of which make living with the condition stressful.
Undergoing a successful gene therapy has the potential to help these patients live a far more normal life. If their bodies produce an adequate level of Factor IX (FIX) on their own, they can reduce the need for or forego these factor replacement therapies altogether. This puts them at less risk of traumatic, uncontrolled bleeding in situations of day-to-day risk of minor injury, serious accidents, surgeries or invasive procedures.
People with moderately severe to severe Hemophilia B usually have FIX levels of <5% before taking the therapy. Two clinical trials followed adult males with moderately severe or severe Hemophilia B who received a single dose. The study participants were then followed for 5 years. Studies have shown an average FIX activity level of 41.5% one year after this gene therapy, and 36.7% two years post-treatment.
Q: How does a patient take this gene therapy?
Dr. Shapiro: A patient is given a one-time infusion of this gene therapy intravenously. The dose is based on the patient’s weight, and the infusion must be given at a specialized hemophilia treatment center like the IHTC. It’s usually given over several hours. The patient will need to be observed for at least 3 hours after the infusion and then make regular follow-up visits to check their factor levels remain adequately elevated and to evaluate their liver function.
Q: How does this new form of gene therapy for Hemophilia B work?
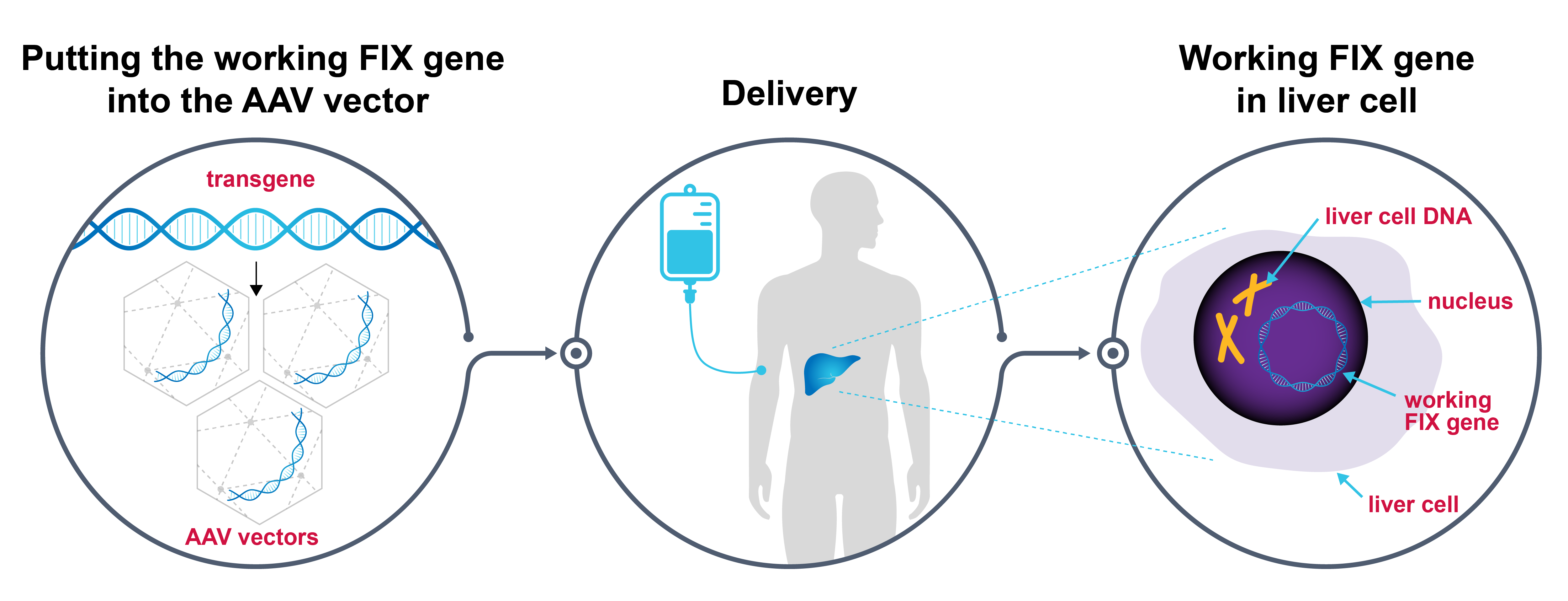
Dr. Shapiro: As mentioned, in patients with Hemophilia B, the gene that makes FIX does not produce a normal level or functional FIX protein. Gene therapy uses a modified virus to deliver a working copy of the FIX gene to the patient’s liver. The delivered FIX gene allows the patient’s liver cells to now produce functional FIX and release it into the blood.
- This gene therapy utilizes a modified virus as a vector (AAV vector) to bring the working copy of the FIX gene into the liver.
- The working FIX gene gets delivered to the patient’s liver cells. It does not become part of their genes in their liver cells and does get placed into their germline cells, meaning that they do not pass this normal FIX gene down to their children.
- The working gene will start making FIX for the patient.
Q: Is this gene therapy a cure for Hemophilia?
Dr. Shapiro: Most people in the studies went from having a severe or moderately severe FIX deficiency to having a mild deficiency, meaning they still had mild Hemophilia B and might still need some treatment at times. FIX activity levels might also go down over time, making it very important to continue close follow-up. These patients should understand that they will still pass on the gene for Hemophilia B to their daughters, even if they are born after treatment with this new form of gene therapy.
Q: Who can take this new form of gene therapy for Hemophilia B?
Dr. Shapiro: Hemgenix® is indicated for the treatment of adults over the age of 18 with Hemophilia B (congenital Factor IX deficiency) without an inhibitor who:
- Currently use Factor IX prophylaxis therapy, or
- Have current or historical life-threatening bleeding, or
- Have repeated, serious spontaneous bleeding episodes
Q: Who cannot take this new form of gene therapy for Hemophilia B?
Dr. Shapiro: Patients with Hemophilia B cannot take this new form of gene therapy if they:
- Are younger than age 18
- Have liver problems
- Have an inhibitor against FIX
- Cannot take steroids (some patients needed steroids for liver irritation)
- Have been exposed to the virus vector in the past and have a high level of immunity to it. These patients will need to discuss this with their doctor, as some people with a past immune response to the virus vector may still have improved FIX levels depending on the level of immunity.
- Cannot come to frequent follow-up visits and lab draws either at or arranged through their hemophilia treatment center, as patients who undergo this form of gene therapy might have to come for more frequent visits and labs to monitor for issues that require intervention.
Q: What are the possible disadvantages of this gene therapy?
Dr. Shapiro: This gene therapy is given as a single infusion, but people who take it will need frequent clinic visits and close monitoring for years after treatment. Possible serious side effects include:
- Liver inflammation: Some people might need to take a steroid for a few months to reverse this damage.
- Becoming immune to the gene therapy carrier (vector). This might prevent use of another gene therapy agent in the future if this treatment fails.
- Possible increased risk of cancer; this has not been observed to date but is discussed due to an inability to estimate very long-term risks.
The most common side effects of this gene therapy are:
- Facial flushing (face getting red and hot)
- Feeling cold
- Shivering
- Rise in blood pressure
- Stomach pain
- Tight feeling in throat
- Itching
- Headache
- Feeling dizzy
- Tight feeling in chest
In clinical trials, 24 out of 54 (44%) people had abnormal liver function tests, and 9 out of 54 (17%) people needed treatment with steroids as a result. On average, these 9 people needed steroid treatment for 80 days.
The actual level of FIX activity might be different for each person. Not everyone will have improvements up to the level that was shown in the studies. This therapy’s use of an AAV vector means that although it doesn’t cause an illness in humans, it is present in the blood and other body fluids after the infusion. Because of this, people who receive this form of gene therapy are not able to donate blood, organs, tissues, or cells for transplant to avoid passing the virus on to other people.